Multi-steps synthesis and total synthesis have been at the center of our concerns for many years. Several total syntheses performed by our team have provided a better understanding of chemical and biological mechanisms.
Total synthesis can therefore be considered as fundamental research. New reactions, cascade sequences, structure elucidations, art and clinical drug targets are strong arguments to maintain an active research in the field of total synthesis of natural products.
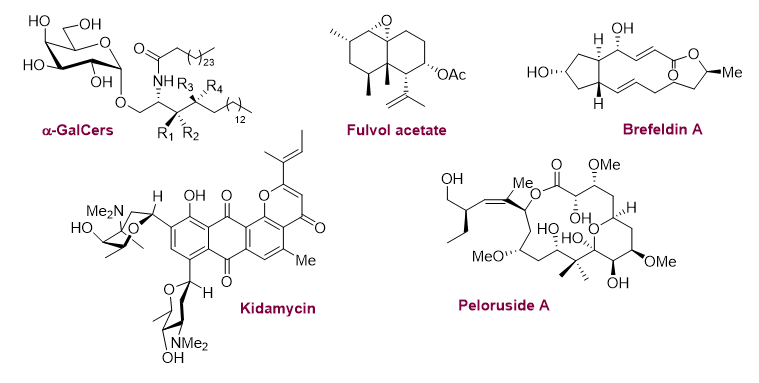
The synthesis of complex molecular structures the development of new methodologies are frequently associated in most of projects developped by our team. Based upon a strong experience in the synthesis of anthraquinonic structures using Diels-Alder approaches, we are studying the total synthesis of kidamycin (ANR KidamySyn 2014-2019).
Beside an original and convergent strategy, we developed a powerful synthetic tool to stereospecifically access a and b aryl-C-glycosides via Mizoroki-Heck cross-coupling of differently substituted glycals with haloarenes.
Team members involved:
- Sylvain COLLET
- Monique MATHÉ-ALLAINMAT
- Muriel PIPELIER
- Jacques LEBRETON
Collaborations
- IMMM - Le Mans, UMR CNRS 6283
- ISCR - Université de Rennes, UMR CNRS 6226
Financial support
- ANR KidamySyn (2014-2019)
- Université de Nantes
Significant references
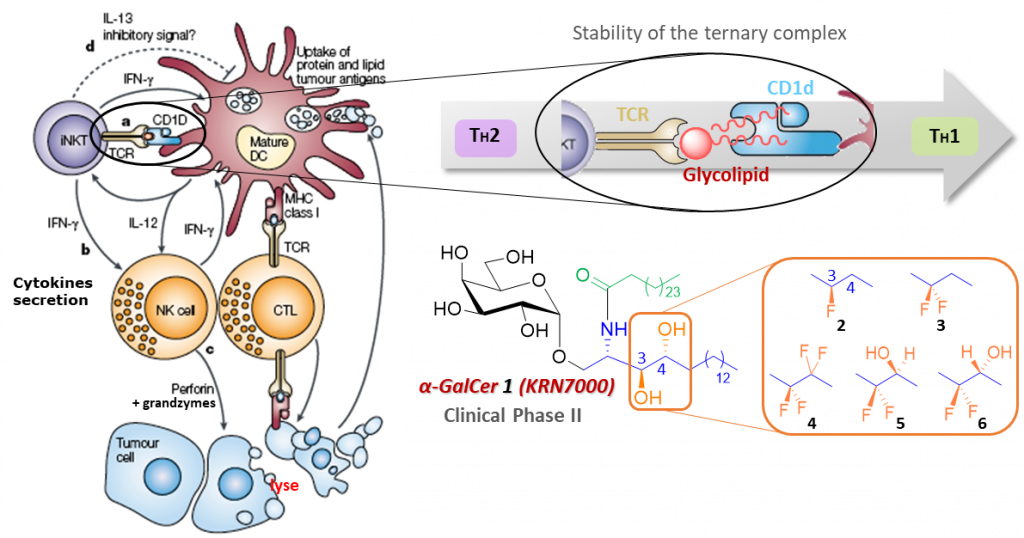
CD1d restricted T lymphocytes, a subclass of lymphocytes, play a pivotal role in the innate-type immune response. A subpopulation of these CD1d restricted lymphocytes, called iNKT cells, feature a semi-invariant T receptor (TCR) that recognizes a variety of glycolipids antigens. In particular, recognition of glycosylceramides bound to CD1d protein receptors of antigen presenting cells (APCs) by iNKT-TCR leads to tertiary complex formation inducing expansion of their population and strong secretion of a large panel of T helper cytokines, including IFN-γ, TNF-α and several interleukins. These cytokines can stimulate the maturation of dendritic cells, activate the production of various cytokines, and stimulate other by-stander immune cells as cytotoxic CD8 lymphocytes. These mechanisms contribute to the inflammatory process, humoral immunity and antibody proliferation depending on two types of helper T cells polarization (TH1 or TH2). It was found that TH1 cytokines (e.g. IFN-γ, IL-2) participate in cell-mediated immunity for tumor rejection and against infections while TH2 cytokines (e.g. IL-4, IL-13) promote auto-immune responses, associated with a variety of diseases such as tuberculosis, type I diabetes, multiple sclerosis and rheumatoid arthritis. Disruption of the TH1/TH2 balance may lead to disease induction as TH1 and TH2 type cytokines can antagonize each other’s biological functions.
Synthetic α-galactosylceramide α-GalCer 1 (also called KRN7000) has been considered as a promising agent against cancer. Recent clinical trials have highlighted its therapeutic potency as a potent adjuvant for vaccines and in anticancer immunotherapy when preloaded on dendritic cells (DCs) or CD1d co-effector, or in combination with programmed cell death 1 (PD-1) blockade proteins.
Combinations of computational and crystal data, with several structure-activity relationship studies on CD1d/α-GalCer analogues/TCR interactions established a relationship between stability of this ternary complex and TH1/TH2 polarisation of the immune response.
Our contribution in this field aims at synthesizing and evaluating new polyfluorinated α-GalCer analogs to study their electronic effect on stability of the ternary complex. Five analogues have been already prepared between 9 and 13 synthesis steps: 3,4-dideoxy-3-fluoro- α -GalCer 2, 3,4-dideoxy-3,3-difluoro- α -GalCer 3, 3,4-dideoxy-3,3,4,4-tetrafluoro-α–GalCer 4, 3-deoxy-3,3-difluoro-a-GalCer 5 and its 4-OH epimer 6.
Team members involved:
- Didier DUBREUIL
- Virginie BLOT
- Muriel PIPELIER
- Arnaud TESSIER
- Jacques LEBRETON
Collaborations
- IMMM - Le Mans, UMR CNRS 6283 : Stéphane GUILLARME,
- Inserm-UMR 1232 CNRS ERL 6001 (Institut de Recherche Santé, CRCINA-IRS) : Jacques LE PENDU.
- Université Southampton (UK) : Bruno LINCLAU.
- Centre René Gauducheau, Centre Régional de Lutte Contre le Cancer Nantes-Atlantique : Jean-Yves DOUILLARD.
Financial support
- ANR PCV GALCERDEO 2008-2012
- ARC et Ligue contre le cancer
- Financement aide Post-doctorants internationaux - Université de Nantes
- Financement d’une thèse – réseau inter-régional transdisciplinaire Glycoouest
Significant references
- JOURNAL OF MEDICINAL CHEMISTRY. 2012, (55), 1227-1241 doi.
- EUROPEAN JOURNAL OF MEDICINAL CHEMISTRY, 2019, (178), 195-213, doi,