While research at the frontier between chemistry and biology is of major importance for the design of new drugs, there is also high potential for developing novel materials for biotechnologies.
In this context, the research activity of the IMF team addresses these two objectives, with, on the one hand, the design of new molecules for cancer treatment by photodynamic therapy, and on the other hand the preparation of innovative materials and nanomaterials for therapy and/or diagnosis. In addition, our interest also focuses on the development of original electrode materials allowing the in-depth investigation of biological mechanisms by electrochemical routes.
Project led by Bruno BUJOLI and Jean-Michel BOULER
Current team members: Bruno Bujoli, Jean-Michel Bouler, Elise Verron, Pascal Janvier, Nathalie Guichard, François-Xavier Lefèvre, Maxence Limelette.
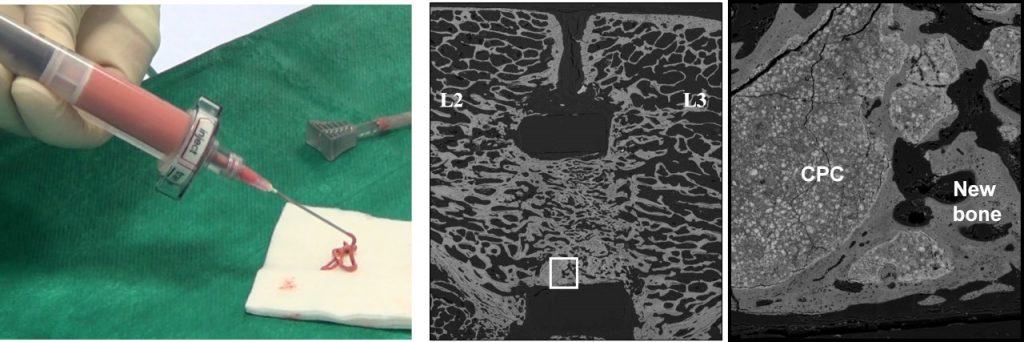
Bone substitutes based on synthetic calcium phosphates (CaP) have been developed for decades, since they can be resorbed in vivo and replaced by natural bone. Their use is now very current in bone surgery, in particular injectable calcium phosphate cements (CPCs) that have appeared as a very promising option, since these biomaterials can be implanted under minimally invasive surgery conditions while offering in addition interesting primary mechanical properties. As a consequence, several brands of injectable CPCs are available on the market for about a decade.
Under a public-private partnership model with the Graftys Company, we are currently developing new generations of injectable CPCs, to address unmet clinical needs.
This includes:
- CPCs combined with drugs in the context of bone therapies, where the CPC will act as a mechanically resistant sacrificial calcium phosphate source for bone reconstruction at the implantation site, and deliver the drug locally, thus producing efficient pharmacological effects only at the specifically intended target sites, with reduced side effects. In this context, bisphosphonates, gallium ions, anaesthetics, water-soluble iodinated aromatic contrast agents have been successfully combined with CPCs formulation and the in vivo behaviour of the resulting compositions has been investigated using animal models.
- Injectable CPCs combined with blood have been developed and were shown to be a promising alternative to autograft for intervertebral arthrodesis. Indeed, the observed fusion rate was found to be at least as good as for the control experiment made using pre-filled cages with autograft. Therefore this strategy might offer a less invasive option for intervertebral spine fusion, allowing a reduction of surgical time, the prevention of morbidity effects due to the bone graft harvesting procedure, and the minimization of the risk of graft loosening and degradation related to the impaction of the pre-filled cage.
Collaborations:
- O. Gauthier (ONIRIS, Nantes),
- Graftys SA (Nantes et Aix-en-Provence),
- Guerbet (Villepinte),
- G. Montavon (UMR 6457 Subatech, Nantes),
- C. Despas & A. Walcarius (UMR 7564 LCPME, Nancy),
- F Fayon, V Montouillout & D. Massiot (UPR 3079 CEMHTI, Orléans),
- J.C. Scimeca (UMR 7277 iBV, Nice),
- COLCOM SA (Clapiers),
- J. Sohier (UMR 5510 MATEIS, Lyon),
- R Debret (UMR 5305, LBTI, Lyon),
- S. Parola et F. Lerouge (UMR 5182, Lab. De chimie de l’ENS, Lyon),
- P. Douek (UMR 5220 /INSERM U1044, CREATIS, Lyon).
Financial supports:
- FUI23, « SpineFlex » project (2017-2020).
- ANR PRCE AAP2018, « BREAKSIT » project (2019-2022).
- CIFRE grant from Graftys (2020-2023).
References:
- Josse, S.; Faucheux, C.; Soueidan, A.; Grimandi, G.; Massiot, D.; Alonso, B.; Janvier, P.; Laib, S.; Gauthier, O.; Daculsi, G.; Guicheux, J.; Bujoli, B.; Bouler, J. M. Chemically modified calcium phosphates as novel materials for bisphosphonate delivery Advanced Materials 2004, 16, 1423.
- Josse, S.; Faucheux, C.; Soueldan, A.; Grimandi, G.; Massiot, D.; Alonso, B.; Janvier, P.; Laib, S.; Pilet, P.; Gauthier, O.; Daculsi, G.; Guicheux, J.; Bujoli, B.; Bouler, J. M. Novel biomaterials for bisphosphonate delivery Biomaterials 2005, 26, 2073.
- Peter, B.; Pioletti, D. P.; Laib, S.; Bujoli, B.; Pilet, P.; Janvier, P.; Guicheux, J.; Zambelli, P. Y.; Bouler, J. M.; Gauthier, O. Calcium phosphate drug delivery system: influence of local zoledronate release on bone implant osteointegration Bone 2005, 36, 52.
- Roussiere, H.; Fayon, F.; Alonso, B.; Rouillon, T.; Schnitzler, V.; Verron, E.; Guicheux, J.; Petit, M.; Massiot, D.; Janvier, P.; Bouler, J. M.; Bujoli, B. Reaction of zoledronate with beta-tricalcium phosphate for the design of potential drug device combined systems Chemistry Of Materials 2008, 20, 182.
- Verron, E.; Gauthier, O.; Janvier, P.; Pilet, P.; Lesoeur, J.; Bujoli, B.; Guicheux, J.; Bouler, J.-M. In vivo bone augmentation in an osteoporotic environment using bisphosphonate-loaded calcium deficient apatite Biomaterials 2010, 31, 7776.
- Schnitzler, V.; Fayon, F.; Despas, C.; Khairoun, I.; Mellier, C.; Rouillon, T.; Massiot, D.; Walcarius, A.; Janvier, P.; Gauthier, O.; Montavon, G.; Bouler, J.-M.; Bujoli, B. Investigation of alendronate-doped Apatitic Cements as a Potential Technology for the Prevention of Osteoporotic Hip Fractures: Critical Influence of the Drug Introduction Mode on the In Vitro Cement Properties Acta Biomaterialia 2011, 7, 759.
- Despas, C.; Schnitzler, V.; Janvier, P.; Fayon, F.; Massiot, D.; Bouler, J.-M.; Bujoli, B.; Walcarius, A. High-frequency impedance measurement as a relevant tool for monitoring the apatitic cement setting reaction Acta Biomaterialia 2014, 10, 940.
- Mellier, C.; Fayon, F.; Boukhechba, F.; Verron, E.; LeFerrec, M.; Montavon, G.; Lesoeur, J.; Schnitzler, V.; Massiot, D.; Janvier, P.; Gauthier, O.; Bouler, J. M.; Bujoli, B. Design and properties of novel gallium-doped injectable apatitic cements Acta Biomaterialia 2015, 24, 322.
- Mellier, C.; Lefevre, F.-X.; Fayon, F.; Montouillout, V.; Despas, C.; Le Ferrec, M.; Boukhechba, F.; Walcarius, A.; Janvier, P.; Dutilleul, M.; Gauthier, O.; Bouler, J.-M.; Bujoli, B. A straightforward approach to enhance the textural, mechanical and biological properties of injectable calcium phosphate apatitic cements (CPCs): CPC/blood composites, a comprehensive study Acta Biomaterialia 2017, 62, 328.
- Le Ferrec, M.; Mellier, C.; Boukhechba, F.; Le Corroller, T.; Guenoun, D.; Fayon, F.; Montouillout, V.; Despas, C.; Walcarius, A.; Massiot, D.; Lefèvre, F.-X.; Robic, C.; Scimeca, J.-C.; Bouler, J.-M.; Bujoli, B. Design and properties of a novel radiopaque injectable apatitic calcium phosphate cement, suitable for image-guided implantation Journal of Biomedical Materials Research Part B: Applied Biomaterials 2018, 106, 2786.
- Dupleichs, M.; Masson, M.; Gauthier, O.; Dutilleul, M.; Bouler, J.-M.; Verron, E.; Janvier, P. Pain Management After Bone Reconstruction Surgery Using an Analgesic Bone Cement: A Functional Noninvasive In Vivo Study Using Gait Analysis Journal of Pain 2018, 19, 1169.
- Le Ferrec, M.; Mellier, C.; Lefèvre, F.-X.; Boukhechba, F.; Janvier, P.; Montavon, G.; Bouler, J.-M.; Gauthier, O.; Bujoli, B. In vivo resorption of injectable apatitic calcium phosphate cements (CPCs): critical role of the intergranular microstructure Journal of Biomedical Materials Research Part B: Applied Biomaterials 2020, 108, 367.
- Gonçalves, H.; Pascal-Moussellard, H.; Lesoeur, J.; Schnitzler, V.; Fellah, B. H.; Pilet, P.; Wagner, N.M.S.; Mellier, C.; Bouler, J-M.; Bujoli, B.; Gauthier O. Injection of calcium phosphate apatitic cement /blood composites in intersomatic fusion cages: a simple and efficient alternative to autograft leading to enhanced spine fusion Spine 2020, in press.
Project led by Fabrice ODOBEL
Cancer still remains one the major cause of mortality in the world in spite of the many progresses made in medical treatments for several decades. Conventional cancer treatment includes surgery, chemotherapy and radiotherapy. Organometallic compounds are attractive weapons against cancers and they have contributed towards the development of efficient modern medicinal anticancer therapies. For examples, the famous cisplatin and its derivatives, carboplatin and oxaliplatin are potent cytotoxic metallodrugs, which are routinely used for decades in clinical treatment for various types of cancer (prostate, testicle, ovary, bladder and lung), but they have dose-limied side effects and some cancer lines have become resistant to these drugs.
Photodynamic therapy (PDT) is an innovative approved clinical treatment in oncology. It has many advantages such as low-cost, less invasiveness and minimal side-effects than conventional chemotherapy. PDT requires a photosensitizer, such as a porphyrin or a phthalocyanine derivative, which upon light excitation produces reactive oxygen species (ROS) in tumor and ultimately leads to cancer cell death, a method that confers higher selectivity to cancer cells than Cisplatin-based chemotherapy treatments.
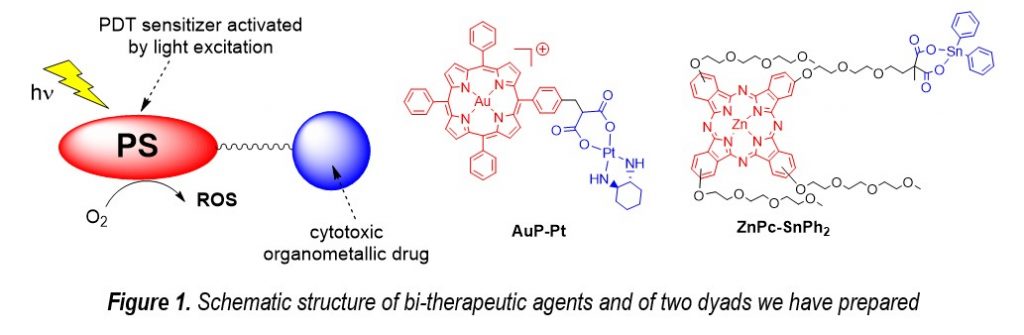
The combination of a PDT sensitizer and an organometallic anticancer drugs could lead to synergetic anticancer properties thanks to the combination of the dark chemostatic effect of the metal complex amplified by the PDT effect generated upon light excitation of the photosensitizer (Figure 1). As a result, a lower dose of drug can be used with lower secondary effects. This approach was the guiding principle to the development of the dyad AuP-Pt which presents an IC50 of 0.9 mM toward MCF-7 cancer cells, whereas LC50 is as high as 1.9 mM for healthy cells.1 Other types of combination including phthalocyanines and tin or gold based anticancer drugs were also designed improve the PDT treatment.
Collaborations:
- Magali GARY-BOBO (Faculté de Pharmacie de Montpellier);
- Céline FROCHOT (Université de Nancy);
- Marwan KOBEISSI (Lebanese University at Beyrouth),
- Prof. Eric VAUTHEY (Université de Genève).
Financial supports:
- Thesis grant from Lebanon CNR and Eiffel fellowship (Campus France).
References:
- I. Toubia; C. Nguyen; S. Diring; L. M. A. Ali; L. Larue; R. Aoun; C. Frochot; M. Gary-Bobo; M. Kobeissi; F. Odobel, "Synthesis and Anticancer Activity of Gold Porphyrin Linked to Malonate Diamine Platinum Complexes." Inorg. Chem., 2019, 58, 18, 12395-12406, https://doi.org/10.1021/acs.inorgchem.9b01981.
Project led by Eléna ISHOW
Nanomaterials nowadays occupy an indisputable place for their diagnostic and therapeutic potentialities, offering new paradigms in the field of medicine in terms of follow-up, safety and personalized treatments. Due to the complexity of the biological environment, mastering the synthesis and understanding the interplay between nanomaterials and biological entities, especially cells, is an imperative need to overcome numerous pitfalls related to toxicity, chemical instability, undesirable targeting, diagnostic artefacts and bioaccumulation, to cite only the major ones.
To this aim, we have been developing for several years multimodal nanoassemblies, originally based on small-molecule photoactive materials, self-assembled as a platform and comprising functional inorganic nanoparticles to bridge the gap from in cellulo to in vivo investigations. Their versatile fabrication offers straightforward architectural tunability, allowing us to decipher their in cellulo interactions.
Particular emphasis is put on the comprehensive description of the physico-chemical and more recently mechanical factors, responsible for the nanoparticle endocytotic cell internalization representing the first compulsory step for drug delivery, labelling of cellular compartments, or remote action to induce cell replication or apoptosis.
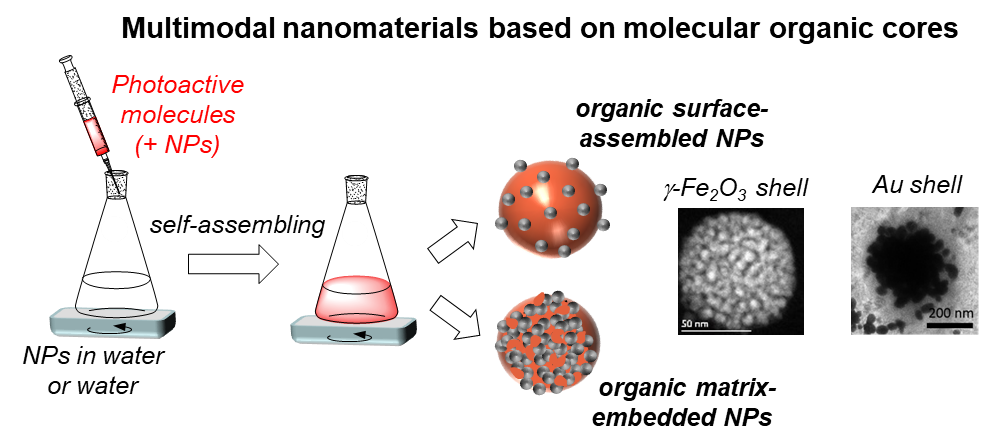
Our group has been developing for more than ten years strong expertise in the synthesis of multimodal nanomaterials originally comprising an organic core made of self-assembled photoactive molecules.
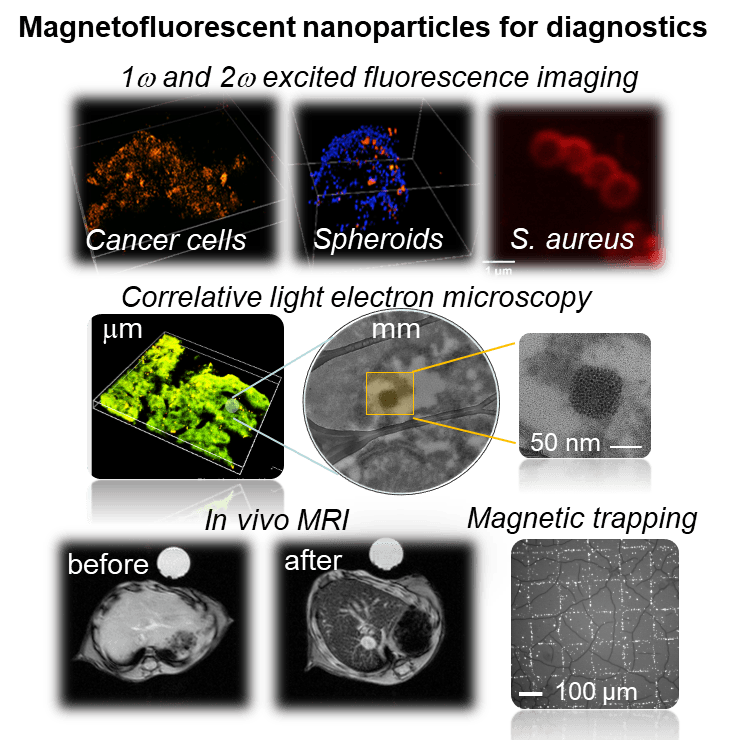
1. Bioimaging using magneto-fluorescent nanomaterials
The fabricated architectures provide easily tunable systems with bright fluorescence associated to gold or magnetic nanoparticles to generate in vivo contrast agents and drug carriers with potential stimulus-driven drug release. Their functionalization with biocompatible agents yields non-cytotoxic nanomaterials, amenable to label various eukaryotic cell lines upon endocytosis (cancer cells, fibroblasts, macrophages) or stain in a minute bacteria walls (S. aureus, E. coli).
Ongoing collaborative efforts with biologists regard the development of properly bioconjugated nanomaterials with proteins or biologically active ligands to specifically target cell receptors for diagnostics and therapy.
2. Microfluidics fabrication
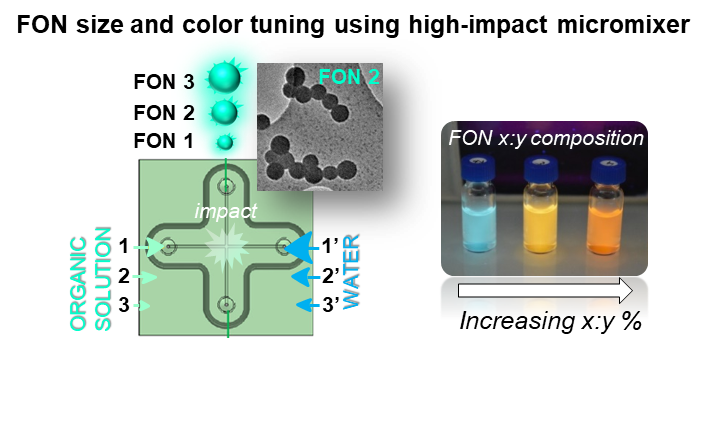
The need for size-controlled nanoparticles represents imperative requirements for metrology and biological use in nanomedicine where the ulterior fate of nanomaterials strongly depends on their dimensions. To this aim, we are currently collaborating with chemical engineers and physicists to synthesize fluorescent organic nanoparticles (FONs) whose size is tuned as a function of the impacting flows and the concentration of the injected organic solution.
Thanks to appropriate compositions, tunable color emission can easily been obtained. Nowadays, our efforts are driven by the modular fabrication of nanoparticles displaying various functional coatings.
3. Mechanobiology
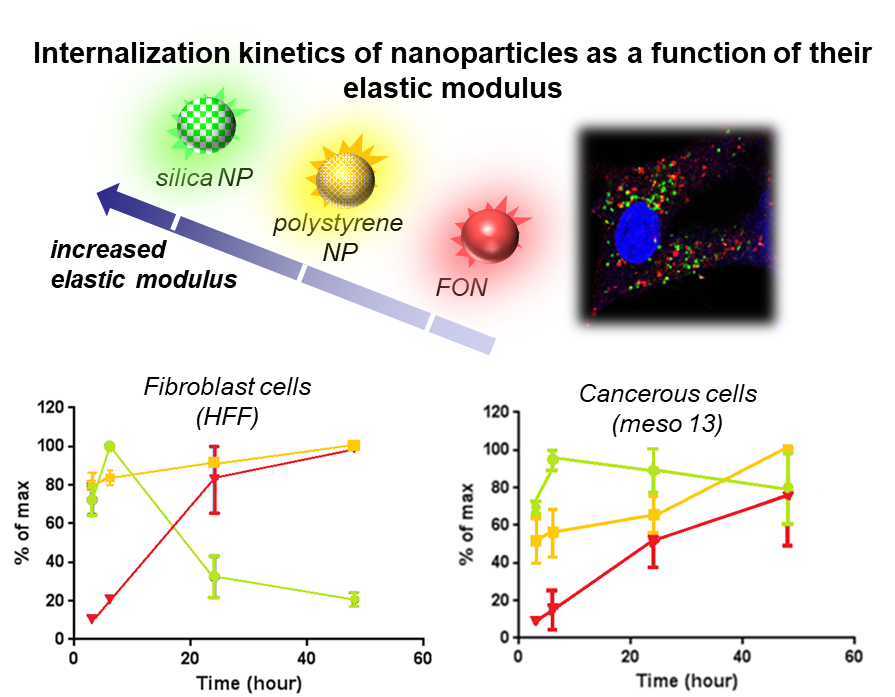
Mechanical issues have scarcely been addressed in biology and medicine although numerous biological phenomena require deep rationalization, like the transport and adhesion of nanomaterials as contrast agents or drug carriers. Through tight collaborations with biologists and physicists, we are using and designing nanomaterials whose mechanical properties can be controlled using light.
This emerging topics has led us to notice differentiate cellular uptake as a function of the elastic modulus of model fluorescent nanoparticles. Currently, we are exploring how photomechanically activable nanoparticles can induce instantaneous cell apoptosis after adhesion upon light irradiation.
Collaborations:
- Christophe Blanquart (CRCINA, Nantes) – Biological fate and impact of nanoparticles for malignant pleural mesothelioma (asbestos cancer)
- Fabrice Fleury, Houda Benhelli-Mokrani (UFIP, Nantes) – DNA repairing and cancer cells
- Stéphanie Sarda et Christelle Combes (ENSIACET, Toulouse) – Apatite and pyrophosphate crystals
- Monique Zagorec (INRA-ONRIS, SECALIM, Nantes) – Bacterial culture
- Philippe Hulin et Steven Nedellec (MicroPICell, SFR Bonamy, Nantes) – Live cell microscopy
- Marc Tramier (IGDR, Université Rennes 1) – Time-resolved microscopy
- Stéphane Cuénot (IMN, Nantes) - Nanomechanics
- Jérôme Bellettre (LTeN, Polytech Nantes) - Microfluidics
- Agnès Montillet (GEPEA, IUT Saint Nazaire, Nantes) – Chemical engineering
- Julien Poly (Université de Haute Alsace, IS2M) – RAFT polymerization
- Jérôme Fresnais (Sorbonne Université, PHENIX) – Magnetic nanoparticles
- Laboratory GeM (IUT Saint Nazaire) – Mechanical modeling
- Sophie Laurent (University of Mons, Belgium) – MRI and relaxometry measurements
References:
- ACS Appl. Mater. Interfaces 11 (36), 32808-32814, 2019 (link)
Boucard, T. Briolay, T. Blondy, M. Boujtita, S. Nedellec, P. Hulin, M. Grégoire, C. Blanquart*, E. Ishow*
Hybrid azo-fluorophore organic nanoparticles emissive turn-on probes for cellular endocytosis. - ACS Omega 3 (12), 17392–17402, 2018 (link)
Boucard, R. Boudjemaa, K. Steenkeste, C. Jacqueline, N. Stephant, F.-X. Lefèvre, A. D. Laurent, L. Lartigue, P. Hulin, S. Nedellec, M.-P. Fontaine, E. Ishow*
Phosphonic acid fluorescent organic nanoparticles for high-contrast and selective staining of gram-positive bacteria. - Small 14 (38), 1802307, 2018 (link)
Boucard, C. Linot, T. Blondy, S. Nedellec, P. Hulin, C. Blanquart, L. Lartigue, E. Ishow*
Small molecule-based fluorescent organic nanoassemblies with strong hydrogen bonding networks for fine tuning and monitoring drug delivery in cancer cells. - Nanoscale 9, 18094-18106, 2017 (link)
Faucon, H. Benhelli-Mokrani, F. Fleury, S. Dutertre, Marc Tramier, J. Boucard, L. Lartigue, S. Nedellec, P. Hulin, E. Ishow*
Bioconjugated fluorescent organic nanoparticles targeting EGFR-overexpressing cancer cells. - Adv. Health. Mater. 4 (17), 2727-2734, 2015 (link
Faucon, H. Benhelli-Mokrani, L. A. Córdova, B. Brulin, D. Heymann, P. Hulin, S. Nedellec, E. Ishow*
Long-term fate of fluorescent organic nanoparticles as bioimaging agents in cancer cells and macrophages.
Funding
- CNRS : Mission Interdisciplinarité (Défi Mécanobiologie - ENAMEL (2018-19), Défi Nano : santé et bien-être - ETHICAM (2014-15))
- Région Pays de la Loire : RFI LUMOMAT - ONASSIS (2016-2019), Paris scientifiques régionaux (partenariat) - NANOTHERAC (2017-2020)
- Université de Nantes : AAP Interdisciplinaires - NANOTRIF (2017), i-site NExT - METCIN (2018-2022)
- Ouest Valorisation
- Ligue contre le cancer
- Campus France : PHC Tournesol - NAPHOMI (2019-2021)
Project led by Mohammed BOUJITA
Current members: Ranil TEMGOUA, Christine THOBIE, Nadine SZUWARSKI, Estelle LEBÈGUE et Mohammed BOUJTITA.

Nowadays, many studies are focusing on various chemical species emerging in the environment whose presence is continuously increasing. These emerging pollutants concern different drugs, personal care products, endocrine disruptors and many other chemicals that are used daily worldwide. In this context, the accumulation of various products in the environment has become a major issue receiving growing attention. In general, the toxicities of some emerging pollutants can be associated with their ability to produce reactive intermediate species by oxidative transformations. Most of the reactive intermediate species are electrophilic intermediates that can react with nucleophilic sites located on proteins.
So when a complex matrix such as environmental samples, the identification and quantification of the transformation products present at low concentrations, often with unknown chemical structures, becomes a greater challenge, so not only the parent compounds but also their intermediate species need to be detected, which requires sophisticated analytical methods.
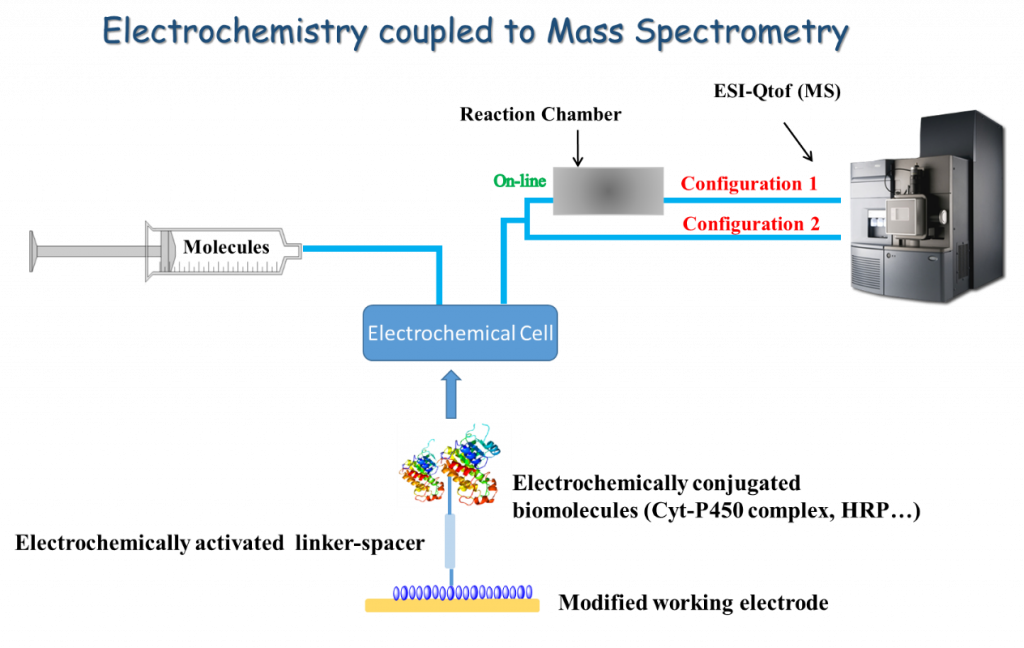
We propose the combination of electrochemical techniques with mass spectrometry technique to evaluate and predict the degradation pathways of various emerging pollutants. In fact, the resulting hybrid technique constitutes a useful tool for predicting various oxidation reactions relevant to oxidative degradations and toxicity aspects. The relevant and innovative points are:
- The reactive intermediate species can easily be identified (species often difficult to identify in complex matrices)
- The degradation pathways can easily be elucidated
- The synthesis of mean products of degradation can be synthesized for further analytical (standards) or biological studies.
Direct oxidative degradations on naked electrodes (Pt, carbon and Au) have been used for the electro-synthesis of a wide range of metabolites or degradation products including N-dealkylation, S-dealkylation, O-dealkylation, N-oxidation, S-oxidation or P-oxidation reactions. However, the use of naked electrodes is restricted and constitutes a useful tool only for reactions initiated by single electron transfer mechanisms (SET).
Therefore, a number of oxidative reactions catalyzed by CYP450s could not be occurred on naked electrodes, including for instance aliphatic hydroxylation reactions that is based on hydrogen atom transfer (HAT) pathways. Our objective is to contribute to the knowledge and development of surfaces modified with a series of CYP450 in order to provide a comprehensive overview of oxidative cleavage pathways.
The modification of electrode surfaces will be carried out by using more selective strategy based on electrochemical Y-click reactions. The resulting strategy can be extended to access the covalent linkage of active compounds used in the field of antibody–drug conjugates, glycoconjugates and drug–drug conjugates. Our research constitutes a science-to-technology approach based on surface modifications, biomimetic structures, bioconjugation and a novel combination of electrochemistry and mass spectrometry/NMR spectrometry.
Financial supports:
- Region Pays de la Loire « MATIERES » project (2013-2017);
- ANR PRC AAP2019 « E-click » project (2019-2023).
References:
- Simulation of the environmental degradation of diuron (herbicide) using electrochemistry coupled to high resolution mass spectrometry; Temgoua, Ranil C.T.; Bussy, Ugo.; Alvarez-Dorta, Dimitri.; Galland ; Hémez, Julie.; Tonlé, Ignas K..; Boujtita, Mohammed (Electrochimica Acta , 2020, 352, 136485),
- Electrochemically Promoted Tyrosine-Click-Chemistry for Protein Labeling; Alvarez-Dorta, Dimitri; Thobie-Gautier, Christine; Croyal, Mikael; Bouzelha, Mohammed; Mevel, Mathieu; Deniaud, David; Boujtita, Mohammed; Gouin, Sebastien G. JOURNAL OF THE AMERICAN CHEMICAL SOCIETY, 2018, 140 (49), 17120-17126,
- Electrochemistry-mass spectrometry to study reactive drug metabolites and CYP450 simulations; Bussy, Ugo; Boisseau, Renaud; Thobie-Gautier, Christine; Boujtita, Mohammed; TRAC-TRENDS IN ANALYTICAL CHEMISTRY, 2015, (70), 67-73,
- In Situ Ultrafast 2D NMR Spectroelectrochemistry for Real-Time Monitoring of Redox Reactions; Boisseau, Renaud; Bussy, Ugo; Giraudeau, Patrick; Boujtita, Mohammed; ANALYTICAL CHEMISTRY, 2015, 87 (1), 372-375,
- Device and method for electrochemically synthesizing intermediate species of a chemical entity; Boujtita, Mohammed ; Bussy, Ugo PCT Int. Appl. (2014), WO 2014108372 A120140717 : China [n° ZL 201480004290.5, 2017] , en Europe [n° 17/43, 2017] et en USA [n° US10385466B2, 2019]
Project led by Estelle LEBÈGUE
Current members: Estelle LEBÈGUE, Christine THOBIE et Mohammed BOUJTITA

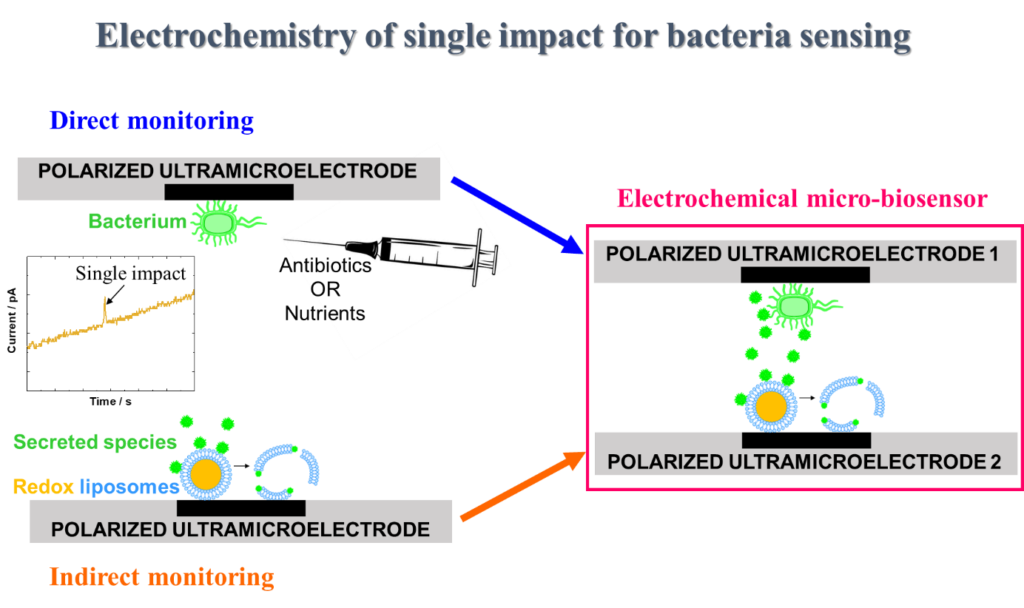
The research project intend to explore the activity and behavior of single (pathogenic) bacterium by monitoring its electrochemical response at an ultramicroelectrode. Beyond the detection and identification of a single cell, the project aims to study one bacterium by monitoring in real time its electrochemical activity in different conditions (presence of antibiotics or nutrients) and under stimulations. The originality of this project is to implement the electrochemical single impact method for sensing and monitoring one bacterium in various environments (under stress or growth conditions).
Two monitoring strategies will be studied and combined:
- Direct monitoring of the bacterial cell through its own electrochemical activity onto the ultramicroelectrode surface.
- Indirect monitoring of the bacterial activity through detection of secreted species with electrochemistry of single redox liposomes nano-impacts.
Our expertise in bio- and nano-electrochemistry, surface modification, biosensors, and recent extension to the electrochemistry of single impact allow us to propose new avenues in electrochemical monitoring of pathogenic bacteria at the single cell scale for micro-biosensor applications.
Financial supports:
- ANR Starting Grant « ELIPOX » (2022 - 2025)
- Région Pays de la Loire « e-NANOBIO » (2021 - 2023)
References:
- Computer-Assisted Processing of Current Step Signals in Single Blocking Impact Electrochemistry
Langlard, Arthur; Smida, Hassiba; Chevalet, Romain; Thobie-Gautier, Christine; Boujtita, Mohammed; Lebègue, Estelle
ACS MEASUREMENT SCIENCE AU, 2024, 4 (5), 585–592, - Exploring the effect of Shewanella oneidensis outer membrane redox proteins in the electrochemical response of single blocking impact events
Smida, Hassiba; Langlard, Arthur; Thomas, Louis; Thobie-Gautier, Christine; Boujtita, Mohammed; Louro, Ricardo O.; Paquete, Catarina M.; Lebègue, Estelle
ELECTROCHIMICA ACTA, 2024, 488, 144235, - Trends in single-impact electrochemistry for bacteria analysis
Smida, Hassiba; Langlard, Arthur; Ameline, Dorine; Thobie-Gautier, Christine; Boujtita, Mohammed; Lebègue, Estelle
ANALYTICAL AND BIOANALYTICAL CHEMISTRY, 2023, 415, 3717, - Single Electrochemical Impacts of Shewanella oneidensis MR-1 Bacteria for Living Cells Adsorption onto a Polarized Ultramicroelectrode Surface
Smida, Hassiba; Lefèvre, François-Xavier; Thobie-Gautier, Christine; Boujtita, Mohammed; Paquete, Catarina M.; Lebègue, Estelle
CHEMELECTROCHEM, 2023, 10 (1), e202200906, - Recent advances in single liposome electrochemistry
Smida, Hassiba; Thobie-Gautier, Christine; Boujtita, Mohammed; Lebègue, Estelle
CURRENT OPINION IN ELECTROCHEMISTRY, 2022, 36, 101141, - Detection of Bacterial Rhamnolipid Toxin by Redox Liposome Single Impact Electrochemistry
Luy, Justine; Ameline, Dorine; Thobie-Gautier, Christine; Boujtita, Mohammed; Lebègue, Estelle
ANGEWANDTE CHEMIE, 2022, 61 (6), e202111416,
